사업분야
Home > 사업분야
사업분야
- Product:
- Vanadyl Sulfate
- Formula:
- VOSO4.XH2O
- CAS No.:
- 27774-13-6
- Product No.:
- SP1024
- Package:
- 25kg card board dum or in terms of customer's requirements.
Description
It is a collection of inorganic compounds of vanadium with the formula , VOSO4(H2O)x where 0≤x≤6. The pentahydrate is common. This hygroscopic blue solid is one of the most common sources of vanadium in the laboratory, reflecting its high stability. It features the vanadyl ion, VO2+, which has been called the "most stable diatomic ion."
Vanadyl sulfate is an intermediate in the extraction of vanadium from petroleum residues, one commercial source of vanadium.
Vanadyl sulfate is an intermediate in the extraction of vanadium from petroleum residues, one commercial source of vanadium.
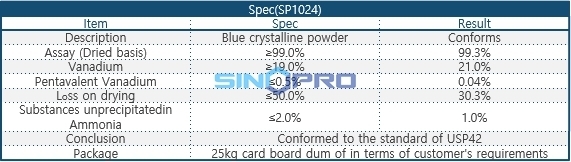
Specification